Nickel is the fifth most common element on earth with both ferrous and nonferrous properties. It is found in lateritic and sulfidic ores which are refined for use in alloys such as stainless steel and high purity nickel suitable for use in manufacturing Lithium-Ion batteries and super alloys.
Elementum Metals: 30/11/2021
Nickel is the fifth most common element on earth with both ferrous and nonferrous properties. It is found in lateritic and sulfidic ores which are refined for use in alloys such as stainless steel and high purity nickel suitable for use in manufacturing Lithium-Ion batteries and super alloys.
Reserves, supply, and processing are concentrated in a handful of countries. Historically stainless steel has been the primary driver of demand; supply of lower grade nickel is not constrained however class 1 nickel is in tight supply; governments concerned about supply security and environmental factors are targeting intensive recycling. Nickel alloys are used extensively in everyday domestic items, infrastructure, transportation, petroleum refineries, chemical manufacturing, healthcare, and space exploration. Nickel is used intensively in EV batteries as it delivers higher energy density and greater storage capacity at a lower cost. It is also used in many other new energy technologies such as wind, nuclear, hydroelectric, hydrogen, solar and geothermal power, as well as carbon capture systems. The prospective supply and demand profile of nickel has led it to be described as the ‘new gasoline’ although the comparison ignores its potential use in a circular economy.
Nickel is a chemical element with the symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge that’s both hard and ductile. It is the fifth most common element on earth and occurs extensively in the earth’s crust and core.1
Nickel was discovered in 15th century Germany as a red-brown ore; because copper could not be extracted it was referred to as Kupfernickel or the Devil’s Copper. Nickel was first identified in 1751 by the Swedish chemist Axel Cronstedt. After the American Civil War nickel and copper were used in coins; ever since which the 5- cent coin has been known as a ‘Nickel’.
Nickel is a transition element with both ferrous properties, such as durability and strength, and nonferrous properties, including high resistance to corrosion, malleability and machinability.
Nickel is found in lateritic ores normally located in tropical regions such as Sulawesi in Indonesia which are mined using open cast methods, and sulfidic ores often found in combination with copper ores such as in Kambalda in the Eastern Goldfields in Western Australia that are mined underground. The northern city of Norilsk is at the centre of Russia’s nickel industry, in the Philippines the Caraga region of Mindanao is highly productive and in Brazil mines are located in the central Goiás region.
Class 2 nickel suitable for use within stainless steel with around 75% purity is obtained from ferronickel (approximately 35% nickel, 65% iron) or nickel pig iron (NPI, between 4 and 13% pure nickel) are mainly processed in China.2
Class 1 nickel in the form of powder, briquettes, electrolytic nickel, or carbonyl nickel, which the London Metal Exchange specifies at least 99.8% purity, such as for use within Lithium-Ion batteries can be obtained from either lateritic or sulfidic ore deposits, although refining lateritic ores generally uses more energy and chemicals than sulfidic.3
Lateritic ores are processed using both hydrometallurgical and pyrometallurgical approaches, the optimal approach depending on the nature of the ore composition. Solvent Extraction – Electrowinning is a hydrometallurgical method using leaching, extractants and electrowinning predominantly applied to lower grade ores. Rotary Kiln Electric Furnace is a pyrometallurgical method that’s suited to ores that are predominantly saprolite involving drying, reduction and smelting. Methods such as the Caron process involve both hydrometallurgical and pyrometallurgical, however costs are high and recovery rates low.
Sulfidic ore is most commonly processed using flash smelting. Dry ore is fed into the furnace along with preheated air, oxygen- enriched air or pure oxygen producing a liquid matte (up to 45% nickel) and slag. The matte containing iron and sulphur is oxidized by injecting air or oxygen into a molten bath.
Various processes are used to refine nickel matte; fluid bed roasting and chlorine- hydrogen reduction produce high-grade nickel oxides with more than 95% nickel, while the carbonyl vapor process is used to produce high-purity nickel pellets.4
The largest nickel producing countries are Indonesia, Philippines, and Russia which together account for 55% of global production. Reserves are concentrated in Indonesia, Australia, and Brazil amounting to 58% of the global total, of which around 60% are lateritic and 40% sulfidic ores.5
Two thirds of global nickel demand is for ferronickel used in the manufacture of stainless steel. Non-ferrous alloys accounts for 12% of global demand, while demand for ferrous alloys accounts for 5%.6
Historically stainless steel has been the primary driver of demand for both class 1 and 2 nickel. Class 1 nickel hit an all-time price high exceeding $50,000 per tonne in 2007 due to high demand exacerbated by low inventories, this was followed by a fall in prices as demand was impacted by the Global Financial Crisis. Expansion in Chinese NPI production in the last decade has led to an increase in class 2 Nickel and corresponding fall in the amount of class 1 used in stainless steel production, causing a decline in class 1 prices. mining companies accordingly scaled back capital expenditure on developing greater production capacity.
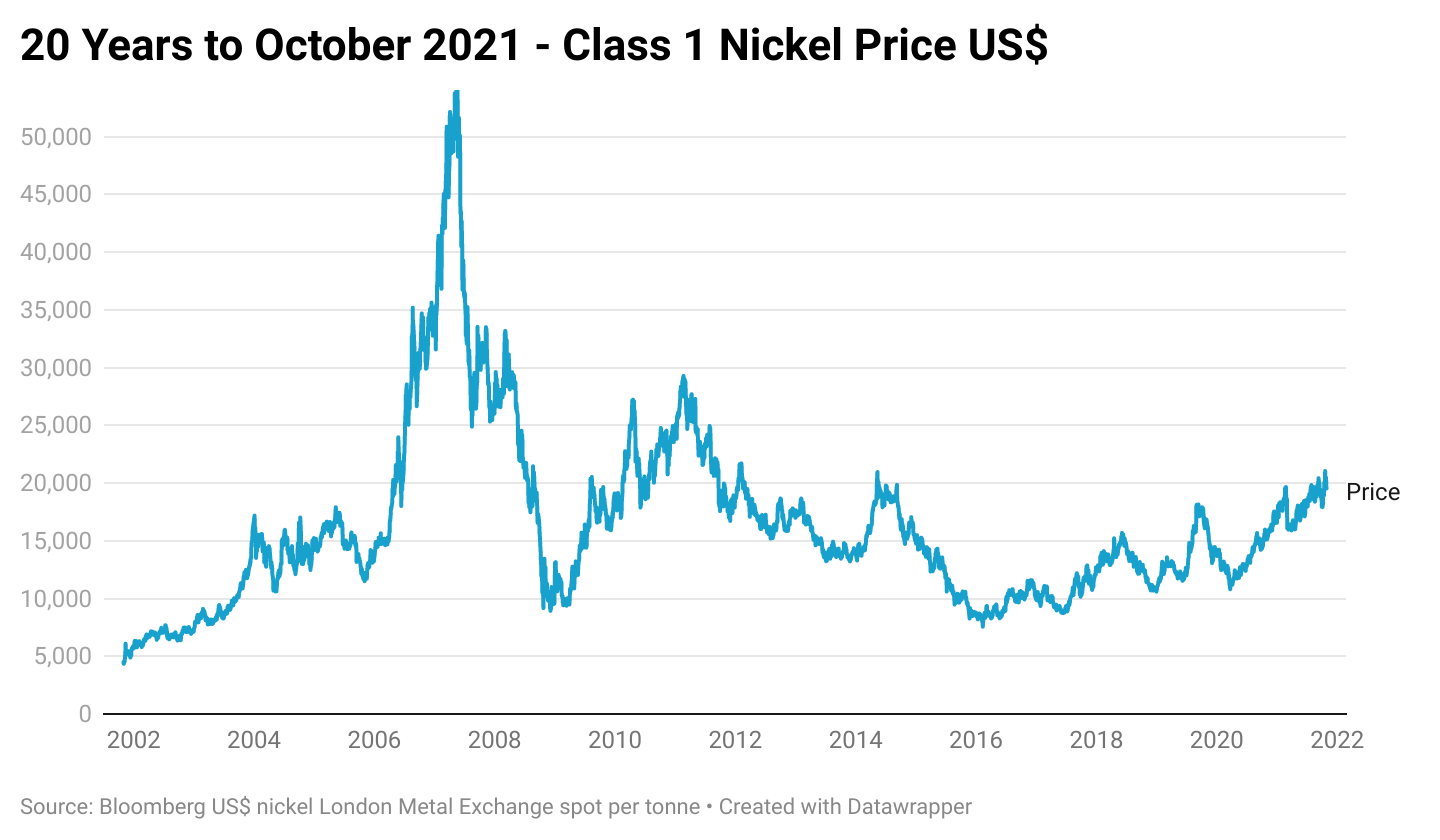
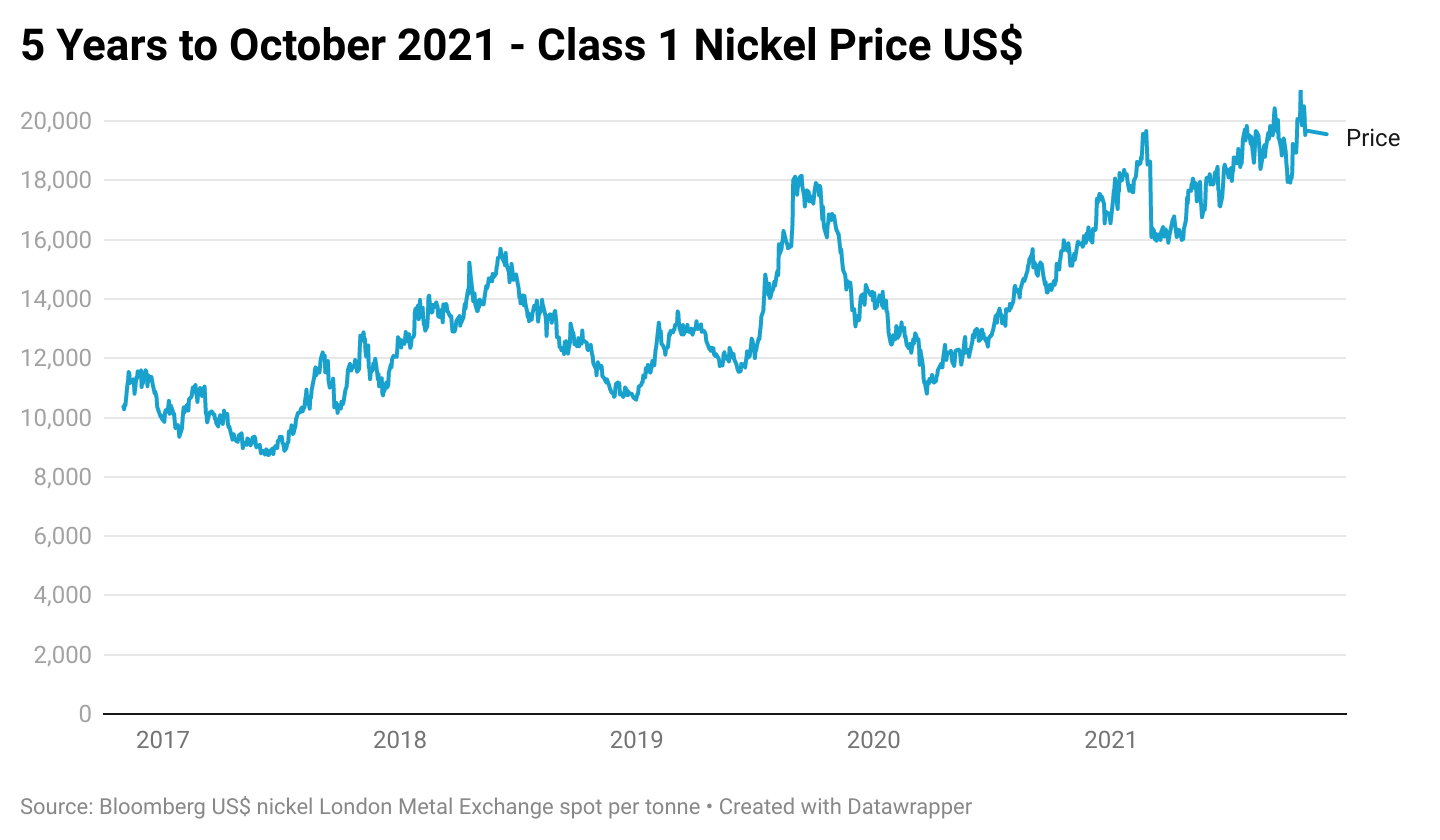
Global demand for nickel is expected to increase by 9.2% to 2.58m tonnes in 2021, roughly split equally between class 1 and class 2, while its estimated supply will increase by 5.8% to 2.64m tonnes. The surplus of 132,000 seen in 2020 is anticipated to fall to 58,000 tonnes.7
While stainless steel stockpiling is widespread and Chinese production of NPI can be rapidly increased in response to price rises, class 1 nickel is in tight supply. Supply constraints of class 1 nickel can be addressed by either reducing class 1 used in stainless steel and other alloys or introducing additional class 1 production capacity.
Tsingshan Holdings Group, one of the world’s largest and most successful stainless- steel producers, in early 2021 announced it had partnered with Huayou Cobalt and CNGR Advanced Material to supply from 2022 onwards 100,000 tonnes of nickel matte, an input for producing nickel sulphate for EV batteries.
The announcement of new class 1 feedstock has caused the nickel price to sink due to the anticipated increase in supply of battery- grade nickel. Even though this may alleviate the class I nickel shortage in theory, the environmental impact of converting NPI to matte may mean that batteries made from this source would not meet environmental standards, essentially quashing the viability of increased supply. Or would require a lot more expenditure to bring it in line with the “cleaner” processes.8
Considering future possible demand and supply dynamics commentators have speculated whether nickel could be the ‘new gasoline’, acknowledging that significant demand growth seems likely while supply is concentrated in a handful of countries with emerging economic status. The comparison highlights the potential for risks to emerge similar to those seen in the 1970s oil crisis.
Recycling
While recycling of stainless steel is well established, in 2019 recycled nickel accounted for 48% of mined production, recycling of class 2 nickel does not directly relieve demand for class 1 for batteries and super alloys.9
Recycling of Lithium-Ion batteries is at an early stage of development however and is regarded by governments such as the EU as vitally important for both supply security and environmental reasons. The EU EV battery strategy includes both financial support for battery manufacturers and recyclers, such as the $400m extended to Northvolt Europe’s leading battery manufacturer, a rigorous recycling regime for vehicle batteries and battery standards including minimum battery lives and durability. The EU framework targets 90% recovery efficiency of nickel from Lithium-Ion batteries by 2025 and 95% by 2030.10
Nickel Alloys
Approximately 3,000 alloys are in use that contain nickel as the principal element, the most common of which is stainless steel. For example stainless steel Type 304 containing 8% nickel is in common use in everyday items such as saucepans, nuts, bolts and domestic sinks, Type 316 containing 11% nickel is employed in more demanding applications such as food processing, brewing and medical applications. Both have an austenitic crystalline structure; Type 304 with good ductility and formability also contains 18% chromium, Type 316 which provides superior resistance to acid and chloride contains 2 – 3% molybdenum.11
Roughly 90% of all new nickel sold every year is used to create alloys such as those used in infrastructure, building construction, transport systems including trains and railway carriages and electricity generation plants. Stainless steel is also widely used in water distribution and treatment and food processing for its non corrosive, non toxic and high hygienic characteristics.
Specialist nickel alloys are used for their superior resistance in highly corrosive environments such as in petrochemical plants as part of wet scrubbing units that reduce sulphurous gas emissions from coal- fired power plants, and in the chemicals industry. Nickel super alloys are used in planes’ jet engines where strength, high temperature tolerance and ability to withstand constant wear is critical.
In healthcare nickel alloys are used within precision instruments such as in robotic surgical devices additionally their non toxic and biocompatibility makes them valued for use in implanted devices such as stents and pacemakers.
Super alloys made with class 1 nickel able to withstand high temperatures and are resistant to oxidation are used in the space industry in rocket motors, space propulsion and power generation. Characteristics including durability in extreme conditions makes them invaluable within satellites and over terrain space vehicles.
Demand for these alloys used within ’old economy’ carbon based economies will continue to grow as developing economies continue to invest in transport infrastructure, construct new cities and develop their industries. Developed economies will also continue to consume these alloys, in particular to revitalise aging infrastructure as does President Biden’s US$1.2trlln Infrastructure Bill approved by the US legislature in early November.
Nickel in the New Economy – Lithium-Ion Batteries
The major advantage of using nickel in batteries is it helps deliver higher energy density and greater storage capacity at a lower cost.Historically a high cobalt content was used to achieve greater energy density however with increasing cobalt prices and concerns about the ethics of cobalt mining, new technology has increasingly focused on the potential of nickel which is now recognised as the key component.
In early battery technologiessuch as the NMC 111 battery, the cathode contained equal quantities of nickel, manganese and cobalt; nickel’s use increased in the NMC 532 and 622, while the most recent high performance NMC 811 battery uses eight parts nickel for each of the other elements.12
The EU’s Joint Research Centre commissioned report from Roskill estimates global nickel demand attributable to batteries will grow to 2.86m tonnes of nickel metal by 2040 of which 95% will relate to automobiles, representing 17.6% per annum compound growth over 20 years. 13
As well as being used in batteries for its electro chemical properties nickel is used to different degrees in many other new energy technologies.

Reliability and long life are critical characteristics in offshore wind farms where there’s a premium on controlling maintenance requirements and costs. Nickel alloys are used for their strength, durability and anti corrosive characteristics: turbine gearboxes require toughness – the ability to withstand mechanical energy without fracturing, while safety equipment must be long lasting and reliable.
Nuclear power plants use nickel alloys within heat transfer and cooling systems as their heat and corrosion resistance are vitally important ensuring durability, reliability and safety. In hydroelectric powerplants the quantity of nickel used is low however its use is critical to ensuring long life of important components such as turbine blades and dam gate controls.
Carbon capture systems collect gas emitted from carbon intensive activities such as coal power stations, transporting gas via either ships or pipes to be sequestered as carbon in rock formations. CO2 is transported in ships at high pressure and low temperature, requiring nickel alloys for their strength and anti corrosive characteristics. Under sea pipelines able to resist corrosion are also used to transport gas to the point of sequestration.
Hydrogen generation involving splitting water molecules into hydrogen and oxygen normally uses rare and expensive catalysts such as platinum. However new techniques are being developed using nickel, reducing costs and increasing efficiency. Green hydrogen is potentially an important source of carbon free energy generated using renewable energy; bulk storage allows it to be used as a replacement for high carbon gasses such as natural gas.
Geothermal power taps natural heat underneath the earth’s surface by capturing energy in water and steam used for heating or generating green electricity.Pipes and component parts are made of nickel alloys for resilience within extreme heat and corrosive environments.
Nickel is used in high specification alloys in solar thermal plants that capture the sun’s rays by conducting heat through molten salt. Metals used in these heat towers need to be resistant to extremely high temperatures and highly corrosive chemicals. Another solar application is nickel’s use in fully transparent solar cells, under development with the intention of turning everyday windows into electricity generating panels.
In conclusion nickel will continue to be widely used both in the ‘old’ economy as well as being an essential component of the new energy revolution. While recycling has historically been focused on alloys, its apparent pure nickel is also well suited to being recycled with the required technology and investment. The limited sources of production create supply risks for many economies, however mandated recycling provides the prospect such risks may be in large part mitigated through the development of circular processing and usage.
http://metalpedia.asianmetal.com/metal/nickel/application.shtml
https://www.lme.com/en/Metals/Non-ferrous/LME-Nickel/Contract-specifications
https://pubs.usgs.gov/periodicals/mcs2020/mcs2020-nickel.pdf
http://metalpedia.asianmetal.com/metal/nickel/application.shtml
https://pubs.usgs.gov/periodicals/mcs2020/mcs2020-nickel.pdf
https://www.europarl.europa.eu/RegData/etudes/BRIE/2021/689337/EPRS_BRI(2021)68933%207_EN.pdf
https://roskill.com/news/nickel-european-commission-publishes-roskill-study/